Did you know that C-nucleosides are essential constituents of modern mRNA vaccines? Were you aware that, up until now, there have been limited options for efficient chemical syntheses? acib changed all that with its recent work (published in Nat.Commun).
Background
The pharmaceutical industry is experiencing a steady increase in demand for C-nucleosides. These valuable compounds serve as essential building blocks for the development of anti-viral agents like remdesivir and anti-tumor treatments. Moreover, the substitution of uracil with the C-nucleoside pseudouridine in mRNA vaccines has been proven effective in enhancing translation efficiency and reducing the immunogenic response to synthetic RNA. Consequently, these modifications have become commonplace in modern mRNA vaccines. However, accessing C-nucleosides and their derivatives has been challenging thus far. Conventional chemical synthesis methods suffer from low reaction efficiencies, requiring cryogenic conditions, the use of protective groups, and toxic chemicals, which results in an expensive and unsustainable process.
Fortunately, our technology offers a solution to overcome these obstacles, providing a promising opportunity for cost-effective and efficient industrial production of this class of substances.
Technology
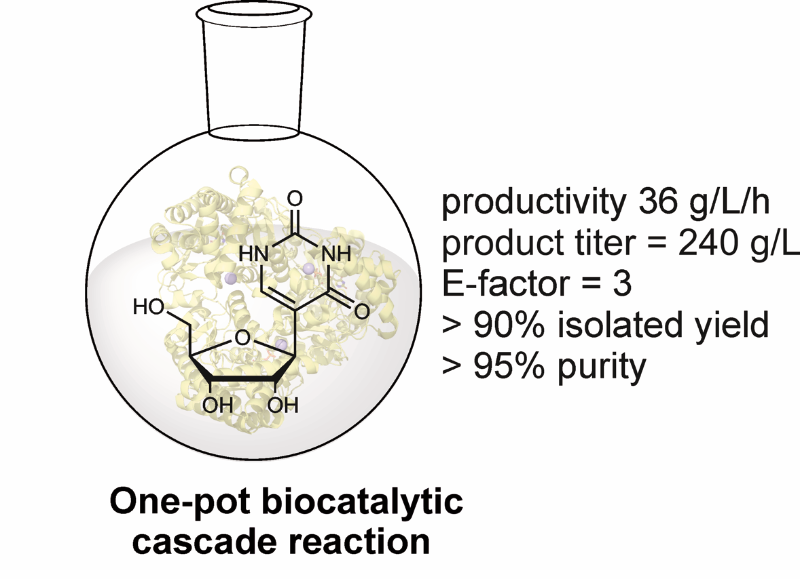
Offer
acib offers to further develop and optimize pseudouridine and other C-nucleoside biocatalysis exclusively for you. IP developed in such projects can be fully transferred to you as our investor/industrial partner.
Experts:
Prof. Dr. Bernd NidetzkyAvailable for:
Joint Research Project, Contract Research, InvestmentsDevelopment status:
Technology Readiness Level 4 (Technology validated in Lab)IPR:
Can be generated for our industrial partners / investorsKeywords:
Biocatalysis, C-nucleoside, One-pot synthesis, Pseudouridine, mRNA vaccines

